India may issue an alert on cough syrup exported by Marion Biotech, whose products have been linked to deaths in Uzbekistan, after tests showed many of the company's drug samples contained toxins.
The Uttar Pradesh Drugs Controlling and Licensing Authority has started the process of cancellation of the manufacturing license of Noida-based Marion Biotech Pvt Ltd after 22 out of 36 samples taken for testing from the firm were found adulterated with Ethylene Glycol.
Ethylene Glycol is a substance linked to the deaths of children who allegedly consumed cough syrup in Gambia, Uzbekistan, and Indonesia.
Indian police on Friday arrested three Marion employees and are looking for two directors after tests in a government laboratory found 22 of 36 syrup samples "adulterated and spurious".As many as 70 children have died in Gambia and 19 in Uzbekistan.
More than 300 children, most under age 5, in Gambia, Indonesia, and Uzbekistan died last year of acute kidney injury associated with contaminated medicines, the WHO said in January.
In addition, it said the Philippines, Timor Leste, Senegal, and Cambodia might be affected because they may have the medicines on sale. It also called for "immediate and concerted action" among its 194 member states to prevent more deaths.
The World Health Organization (WHO) issued a product alert in January as "Two cough syrups made by India's Marion Biotech should not be used for children after the products were linked to 19 deaths in Uzbekistan".
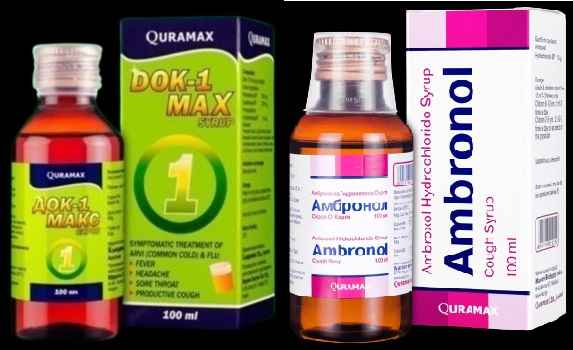
Indian local investigating body has submitted its report to the health ministry and the health ministry is expected to issue an alert for Ambronol and DOK-1 Max syrups which were linked to the deaths of 18 children in Uzbekistan.